Bacterial microcompartments
Bacterial microcompartments (BMCs) are composed of selectively permeable proteinaceous shells and their encapsulated enzymatic cargo. BMCs encapsulate at least two enzymes, which catalyze sequential reactions of a given metabolic pathway. Similarly to membrane-bound organelles in eukaryotes, the selectively permeable BMC shells sequester enzymes and reactants in a compartment distinct from the remainder of the cytosol, thereby increasing local concentrations, reducing unwanted side-reactions, and preventing escape of volatile reaction intermediates. BMCs are wide-spread, having been detected in more than 20 bacterial phyla, and can be subdivided into two major groups: Carboxysomes are filled with RuBisCo and occur in photosynthetic cyanobacteria, where they contribute to a carbon cencentrating mechanism that enhances photosynthesis, and in some chemoautotrophic bacteria. Metabolosomes, such as the BMC of the marine myxobacterium Haliangium ochraceum, are typically involved in aldehyde metabolism and shield their host from toxic intermediates. Some pathogenic bacteria use metabolsome BMCs to catalyze rections that provide them with a competitive growth advantage. Our work on BMCs resulted from a collaboration with Markus Sutter and the laboratory of Prof. Cheryl Kerfeld at the Lawrence Berkeley National Laboratory (LBNL).
The crystal structure of the HO BMC shell: Architecture and assembly principles
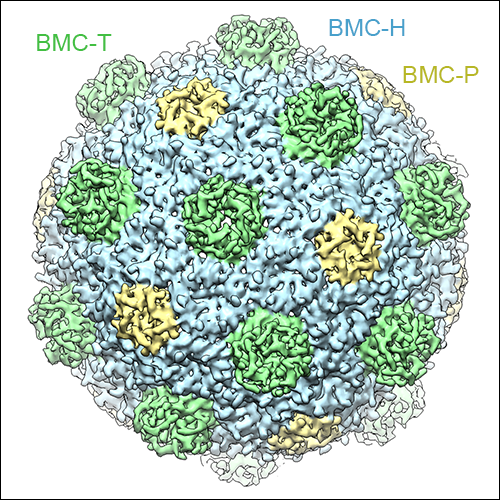
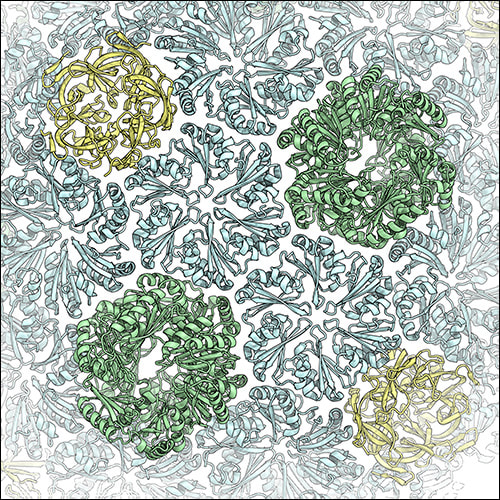
BMCs shells are formed by characteristic shell proteins that harbor the so-called BMC-fold, which assemble into hexamers (BMC-H), trimers (BMC-T) or pentamers (BMC-P). Selective exchange of metabolies between the interior of the shell and the cellular metabolism occurs through pores at the center of these oligomeric assemblies. Additional sub-types of shell proteins exist: The BMC shell from H. ochraceum contains three BMC-T variants, two of which assemble into stacked dimers of trimers that harbor a large central cavity that can be accessed through gated pores. While the structures of these individual shell protein oligomers had been been determined, the structure of an intact BMC shell remained elusive until Markus Sutter in the laboratory of Prof. Cheryl Kerfeld managed to crystallize the H. ochraceum BMC shell. In order to solve the crystal structure, we first obtained a sub-nanometer resolution cryo-EM reconstruction of the recombinantly expressed H. ochraceum BMC shell, into which previously determined high-resolution structures of BMC shell components could be docked. This structural model could then be used as a molecular replacement search model for phasing of X-ray diffraction data from crystals containing the intact shell. This phasing approached proved to be successful and enabled the building of a complete atomic model of the H. ochraceum BMC shell at 3.5 Å resolution. This structure revealed for the first time the architectural principles according to which the individual components assemble to form a complete BMC shell: The BMC-H subunits form the facets of the shell, while the BMC-P modules occupy the pentameric vertices of the icosahedral assembly, and the BMC-T subunits are found at the three-fold symmetry axes.
|
Left: The cryo-EM reconstruction of the HO BMC shell at sub-nanometer resolution. BMC-H subunits are shown in blue, BMC-P in yellow, and BMC-T in green. The double-stacked BMC-Ts can be seen to form protrusions on the shell surface. Right: Close-up view of the X-ray crystal structure of the HO BMC shell.
Sutter M, Greber B, Aussignargues C, Kerfeld CA (2017). Assembly Principles and Structure of a 6.5-MDa Bacterial Microcompartment Shell. Science 356 (6344): 1293-1297. PubMed F1000Prime |
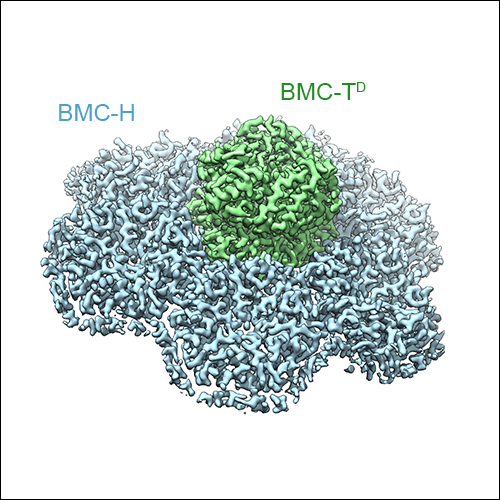
The cryo-EM structure of the HO BMC shell: Higher resolution and asymmetric analysis
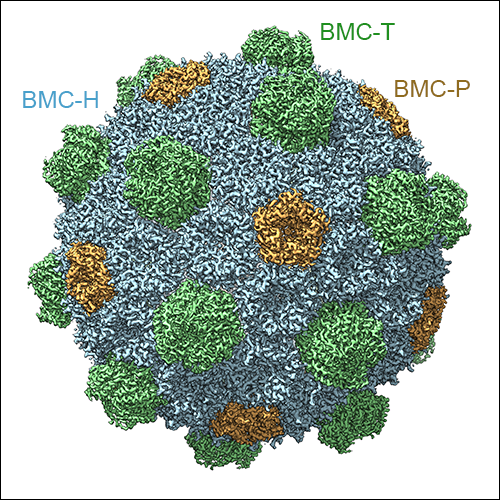
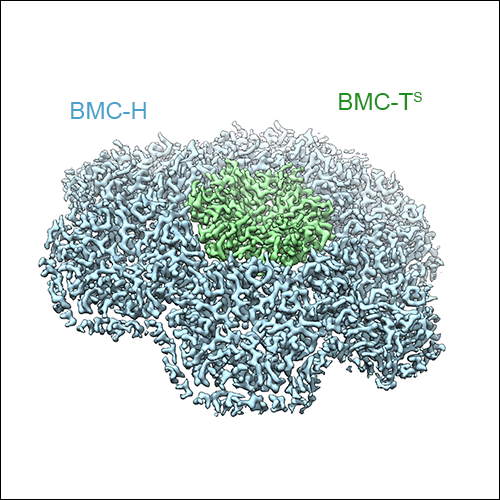
As mentioned above, three types of BMC-T subunits exist in the HO BMC shell, two of which form double-stacked dimers of trimers, while one type forms regular trimeric assemblies. Due to averaging inherent X-ray diffraction from crystals, these variants could not be distinguished in the X-ray crystal structure of the HO BMC shell. However, cryo-EM data preserves all the information from all the asymmetric units in the raw micrographs, and therefore these structural features could be resolved using an asymmetric cryo-EM data processing strategy. We were able to obtain a symmetric reconstruction of the HO BMC shell at 3.0 Å resolution, as well as cryo-EM maps showing the double- and single-stacked BMC-T assemblies in the context of the assembled shell. Additionally, we were able to obtain insight into the conformational variability of the entire shell, and of the gated pores of the double-stacked BMC-T variants.
Left: The high-resolution cryo-EM reconstruction of the HO BMC shell at 3 Å resolution. BMC-H subunits are shown in blue, BMC-P in orange, and BMC-T in green. The double-stacked BMC-Ts can be seen to form protrusions on the shell surface. Middle: Sub-volume classified for presence of a single-stacked BMC-T trimer. Right: Sub-volume classified for the presence of a double stacked BMC-T dimer of trimers.
Greber BJ*, Sutter M*, Kerfeld CA (2019). The Plasticity of Molecular Interactions Governs Bacterial Microcompartment Shell Assembly. Structure 27 (5): 749-763.e4. *equal contribution PubMed
Greber BJ*, Sutter M*, Kerfeld CA (2019). The Plasticity of Molecular Interactions Governs Bacterial Microcompartment Shell Assembly. Structure 27 (5): 749-763.e4. *equal contribution PubMed