Eukaryotic ribosome biogenesis
Eukaryotic ribosomes are more complex than their bacterial counterparts. Correspondingly, the assembly of eukaryotic ribosomal subunits in the cell is a highly intricate and regulated process requiring the activity of several hundred so-called trans-acting factors. During his doctoral and post-doctoral studies in the laboratory of Prof. Nenad Ban at ETH Zurich, Basil Greber aimed to elucidate how ribosome biogenesis factors bound near the ribosomal polypeptide tunnel exit contribute to nuclear export and cytoplasmic maturation of the pre-60S particle.
|
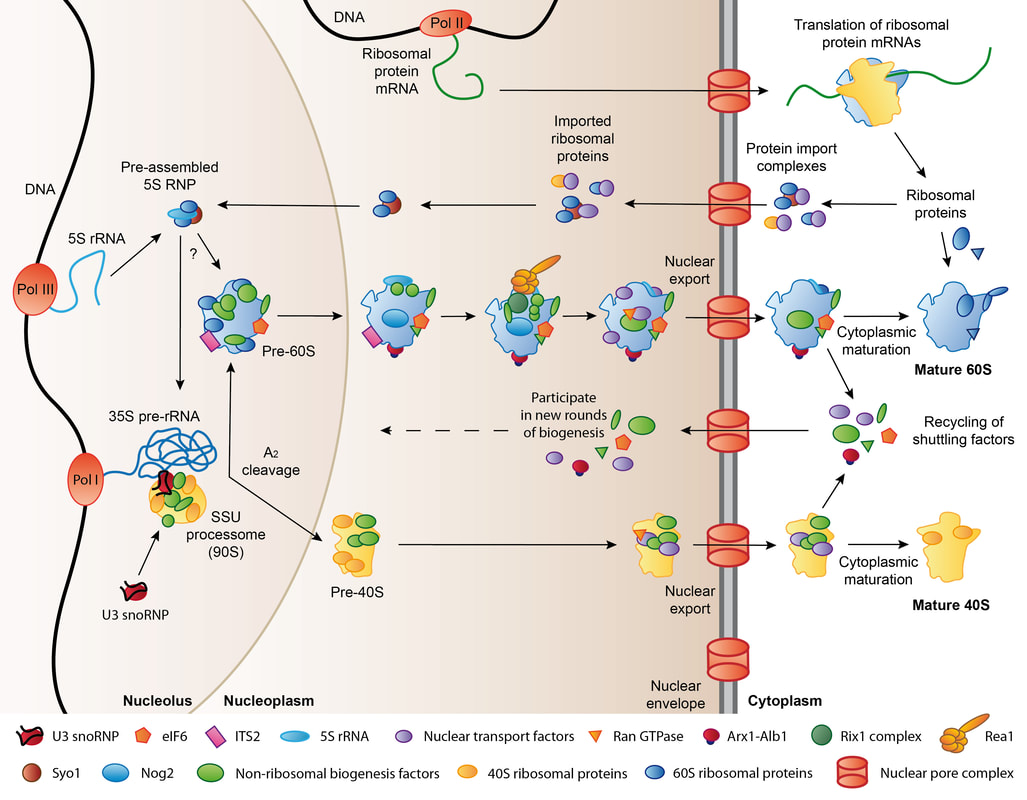
Schematic overview of ribosome biogenesis in eukaryotes. Ribosome biogenesis starts in the nucleolus and then proceeds though nucleoplasmic and cytoplasmic maturation steps. A large number of trans-acting ribosome biogenesis factors is required for assembly, nuclear export, and maturation of the pre-ribosomal particles. Figure originally published in Greber (2016), RNA 22 (11): 1643-1662.
Greber BJ (2016). Mechanistic Insight into Eukaryotic 60S Ribosomal Subunit Biogenesis by Cryo-Electron Microscopy. RNA 22 (11): 1643-1662. PubMed |
The cryo-EM structure of the 60S-Arx1-Rei1 complex
|
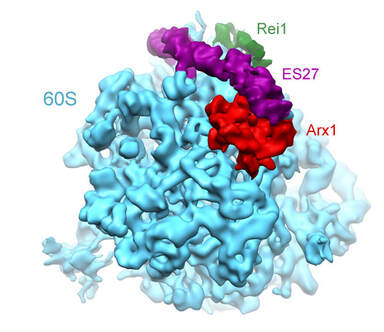
Cryo-EM reconstruction of the 60S-Arx1-Rei1 complex at 8.1 Å resolution. Arx1 (red) binds to the 60S subunit (blue) near the polypeptide tunnel exit and also stabilizes the eukaryotic-specific expansion segment ES27 (purple).
Greber BJ, Boehringer D, Montellese C, Ban N (2012). Cryo-EM structures of Arx1 and maturation factors Rei1 and Jjj1 bound to the 60S ribosomal subunit. Nat. Struct. Mol. Biol. 19 (12): 1228–1233. PubMed |
Using cryo-electron microscopy (cryo-EM), we were able to obtain insight into the interactions of the pre-60S nuclear export factor Arx1 with the 60S ribosomal subunit at sub-nanometer resolution. This structure showed that Arx1 sits above the ribosomal tunnel exit, with a cavity previously implicated in interactions with components of the nuclear pore facing the ribosomal surface and mostly shielded from the outside. Additionally, Arx1 interacts with the long eukaryotic rRNA expansion segment 27 (ES27) and thereby structurally stabilizes this long helical RNA structure. Our structures also revealed the ribosomal binding sites of the cytoplasmic maturation factors Rei1 and Jjj1, which have been implicated in Arx1 release from the pre-60S particle.
The high-resolution cryo-EM structure of the 60S-Arx1-Alb1-Rei1 complex: Rei1 probes the ribosomal tunnel
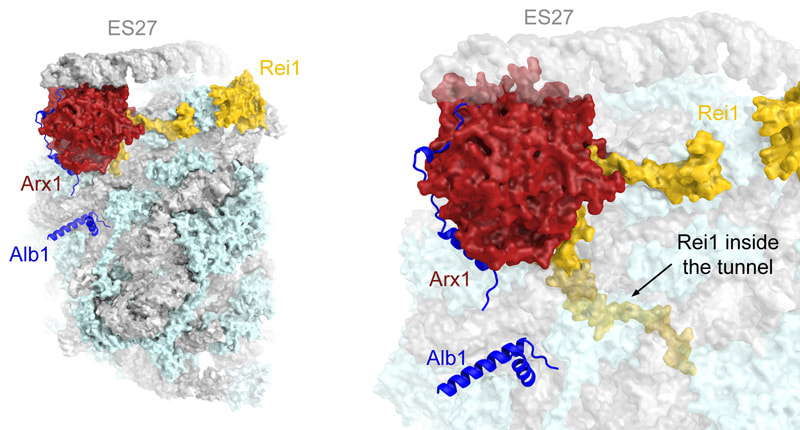
Based on cryo-EM data collected on a Falcon II direct electron detector and using a preparation of an Alb1-Arx1 complex provided by the laboratory of Nicolas Leulliot (CNRS Paris), we were able to determine the structures of in vitro-reconstituted 60S-Arx1-Alb1-Rei1 and 60S-Arx1-Rei1 complexes to near-atomic resolutions of 3.4-3.9 Å. These structures allowed us to build an atomic model for Arx1 including Arx1-specific extensions that are involved in interactions with the 60S subunit. Furthermore, aided by chemical crosslinking-mass spectrometry data obtained by Alexander Leitner in the laboratory of Ruedi Aebersold (ETH Zurich), we were able to identify the position of Alb1 in the complex. Strikingly, these structures also revealed that Rei1 is deeply inserted into the polypeptide tunnel from the exit site, reaching to within 15 Å of the peptidyl transferase active site at the other end of the tunnel. An in-depth biochemical and yeast genetics analysis performed by Stefan Gerhardy in the laboratory of Vikram Panse (ETH Zurich; now at the University of Zurich) revealed that failure of Rei1 to properly insert into the polypeptide tunnel leads to ribosome biogenesis defects in yeast, including formation of half-mers and mis-localization of late cytoplasmic maturation factors such as Arx1 and eIF6. This suggests that Rei1 probes the ribosomal tunnel and that perturbations of the tunnel preventing insertion lead to an arrest of cytoplasmic maturation.
|
Rendering of the molecular structure of the 60S-Arx1-Alb1-Rei1 complex determined at 3.4 Å resolution by cryo-EM. Alb1 (blue) interacts with Arx1 (red) and the 60S subunit. Rei1 inserts its C-terminus deeply into the ribosomal polypeptide tunnel exit (60S subunit rendered semi-transparent), thereby ensuring that the tunnel of the nascent pre-60S particle is not blocked.
Greber BJ*, Gerhardy S*, Leitner A, Leibundgut M, Salem M, Boehringer D, Leulliot N, Aebersold R, Panse VG, Ban N (2016). Insertion of the Ribosome Biogenesis Factor Rei1 Probes the Ribosomal Tunnel during 60S Maturation. Cell 164 (1-2): 91–102. *equal contribution. PubMed |
For additional information on eukaryotic ribosomes, including PyMol scripts for structure visualization and a complete guide to the nomenclature of ribosomal proteins, please visit the website of the laboratory of Prof. Nenad Ban at ETH Zurich.